Lumibird Medical accelerates Asia-Pacific expansion with future acquisition of BIZ MEDIC SDN BHD to form new Southeast Asian hub

Lumibird Medical today announced that it has entered into discussions to acquire its long-term distribution partner, BIZ MEDIC SDN BHD, based in Kuala Lumpur, Malaysia. This move marks the future establishment of Lumibird Medical Southeast Asia, a new hub designed to serve as the group’s primary engine for growth in the world’s most high-potential ophthalmology market.
ICL sizing assistance with ABSolu ultrasound platform compatible with ICL Guru

Lumibird Medical is proud to announce the launch of a new option for its cutting-edge ABSolu® ultrasound platform, now compatible with ICL Guru (REVAI platform) for Implantable Collamer Lens (ICL) sizing.
Installation of the first clinical prototype featuring HoloDoppler technology
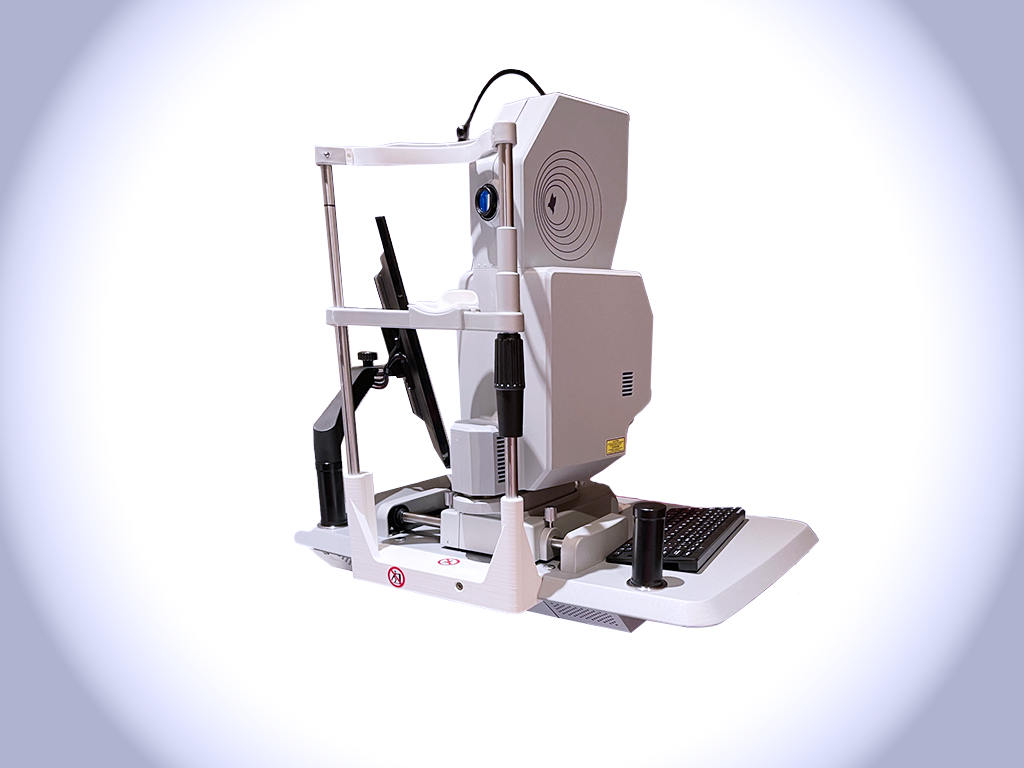
Lumibird Medical has installed the first clinical prototype of its Doppler holography system at the Hôpital National des Quinze-Vingts.
Lumibird Medical rebrands its Japanese subsidiary to reflect strategic growth

Lumibird Medical is pleased to announce the rebranding of its Japanese subsidiary. Formerly known as Ellex Japan, the company will now operate under the name Lumibird Medical Japan effective immediately.
POCKET III™, The new handheld pachymeter

Light, connected and safe are the keywords of POCKET III™, the next-generation handheld pachymeter from Quantel Medical, by Lumibird Medical, the world leader in ophthalmic ultrasound.
C.DIAG® – Dry eye diagnostic aid platform
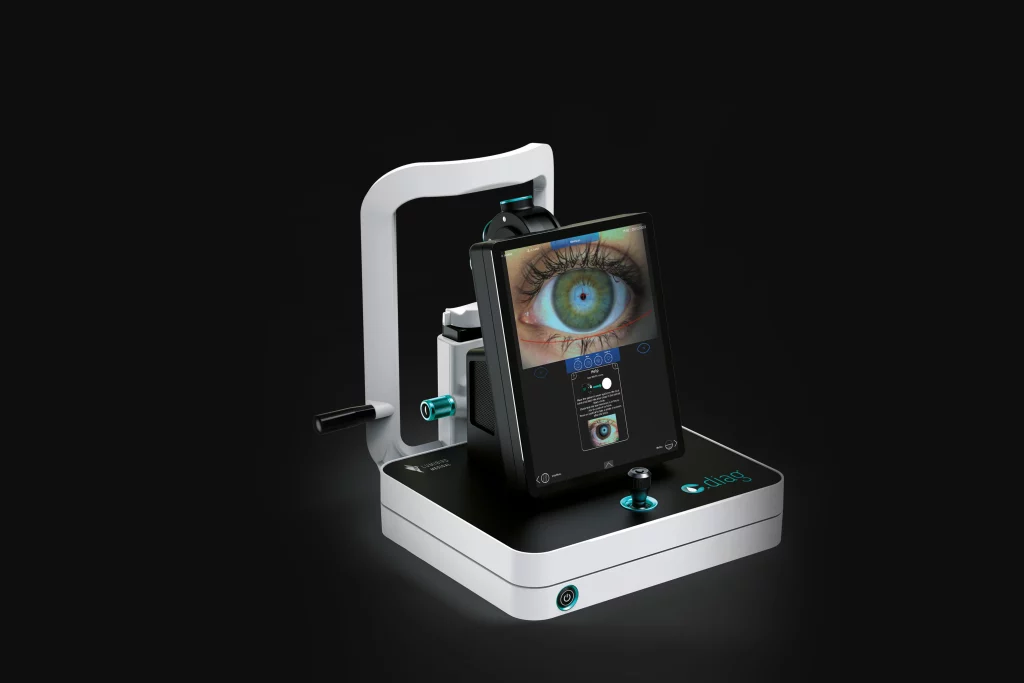
Lumibird Medical announces the launch of the C.DIAG®, a new-generation dry eye diagnostic aid platform with integrated AI algorithms.
Quantel Medical one of the first winners of the France 2030 “Medical Imaging” call for projects.

Lumibird Medical® is announcing that
the HoloDoppler project led by its subsidiary Quantel Medical has been selected as part of the Health section of France 2030, financed by Bpifrance.
Lumibird Medical® Polska

Lumibird Medical® Group renames its Polish subsidiary to Lumibird Medical® Polska, which moves to new offices in Warsaw to support its growth in the local market.
Neo Premium Laser range by Lumibird Medical

Lumibird Medical announces the launch of their two next generation Nd:YAG/SLT lasers, the Tango Neo and UltraQ Reflex Neo.
Lumibird Medical receives marketing approval in China for its Vitra 2 Photocoagulator

Lumibird Medical is proud to announce that it has received marketing approval in China for its Vitra 2 photocoagulation laser from the Chinese Health Authority (NMPA).